The engineering of chimeric antigen receptor (CAR) T cells has been a significant recent advance in cancer therapy. CAR T cells are manufactured by collecting a patient’s own T cells, engineering these T cells to express a synthetic signaling molecule (the CAR) that specifically redirects the T cell response towards cancer cells, and re-infusing the engineered cells back into the patient.
CAR T cell therapies have shown remarkable success in treating liquid cancers such as lymphomas and leukemias, with one dose of CAR T cells directed towards the B cell antigen CD19 resulting in a long-lasting response in nearly half of patients with relapsed or refractory B cell lymphomas1. However, these blood cancers only account for about 7% of deaths among 36 different cancers2, and CAR T cell therapies have yet to exhibit similar efficacy in solid tumors3.
Given the potential of CAR T cells, significant focus has been placed on developing engineering strategies to boost the function of these cellular therapies. Cytokines, or signaling molecules used by immune cells to communicate with each other, may be key to augmenting T cell function. Common gamma chain cytokines such as IL-2, which utilize a protein subunit shared between several cytokines, have been long recognized for their potential to enhance CAR T cells due to their important role in regulating T cell differentiation and function. However, systemic administration of these cytokines can often do more harm than good, with therapeutic doses of these powerful cytokines causing intolerable systemic inflammation and dose-limiting toxicity.
In this work, Jiang et al. seek to characterize the effect of another relatively less well-characterized gamma chain cytokine, IL-9, on CAR T cell function.
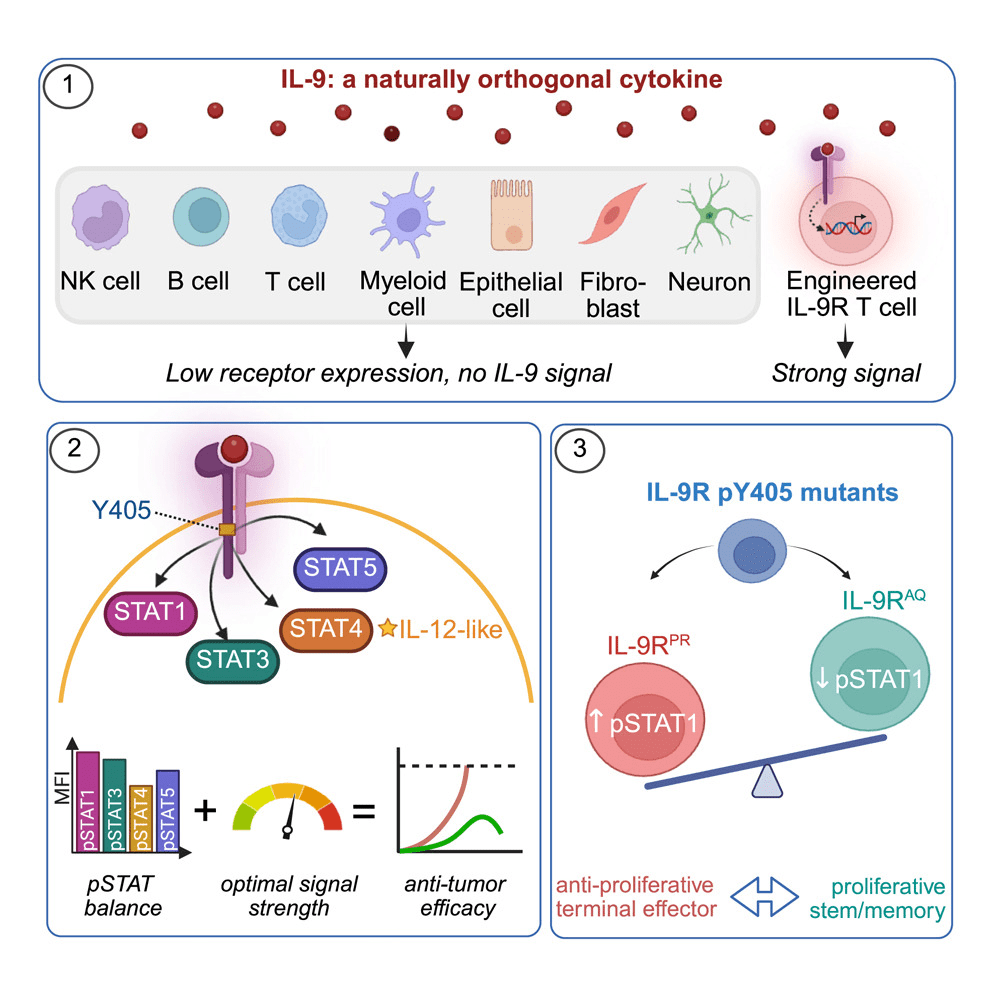
Using a variety of genomic, transcriptomic, and functional approaches, the authors found that the IL-9 receptor (IL-9R) is extremely lowly expressed in immune and normal tissues, hinting at the favorable safety profile of therapeutic high dose IL-9 treatment. When treating CAR T cells that have been engineered to express IL-9R with IL-9, these CAR T cells showed better tissue infiltration and stemness, characteristics associated with better anti-tumor response. Importantly, these IL-9 treated CAR T cells demonstrated enhanced tumor clearance in mouse models without causing widespread systemic inflammation and toxicity.
Common gamma chain cytokines signal through the JAK-STAT pathway which allows for rapid membrane-to-nucleus signaling. Each cytokine recruits a unique combination of STAT proteins, leading to the activation of specific gene programs downstream. Signaling through the IL-9R complex recruits STAT1, STAT3, STAT5, and interestingly, STAT4. STAT4 is typically not thought to be associated with gamma chain cytokine pathways, but instead with IL-12 receptor signaling. High-dose IL-12 administration has been shown to have anti-tumor activity; however, systemic dosing of this potent pro-inflammatory cytokine results in severe and dose-limiting toxicity4. The natural orthogonality of the IL-9/IL-9R axis raises the possibility of exploiting the anti-tumor potential of IL-12 signaling without the systemic toxicities associated with IL-12 treatment.
Small mutations in IL-9 or IL-9R can elicit changes in various parts of the JAK-STAT pathway, tuning the T cell response to the cytokine stimulus. Attenuating or amplifying signaling through the structural changes in the IL-9R complex weakens T cells’ ability to control tumor growth. Furthermore, adjusting the distribution of the STAT proteins recruited by IL-9 prompts changes in T cell proliferation and tumor infiltration, but ultimately does not result in improved anti-tumor efficacy. These data suggest that both the native signaling strength and downstream signaling cascade through IL-9R confers the best IL-9-induced anti-tumor response.
When evaluated in models of human hematological and solid cancers, human CAR T cells equipped with the IL-9 receptor and treated with IL-9 demonstrated a clear therapeutic advantage compared to unmodified CAR T cells. Specifically, these IL-9R-expressing CAR T cells slowed tumor growth in an osteosarcoma model and cleared the cancer entirely in an acute lymphoblastic leukemia model.
Jiang et al., demonstrates that the natural orthogonality conferred by the restricted expression profile of IL-9R across immune and normal cell types is suggestive of a favorable safety profile. Signaling through the native IL-9R complex recruits STAT1, STAT3, STAT5, and surprisingly, STAT4 activity. Furthermore, changes to IL-9 or IL-9R resulting in alterations in the downstream signaling cascade components or signaling strength attenuates the anti-tumor function of IL-9R-expressing T cells treated with IL-9. CAR T cells equipped with the IL-9/IL-9R signaling axis demonstrated improved anti-tumor activity in human models of solid and liquid cancers.
Together, the authors illustrate the potential of harnessing the IL-9 signaling cascade to improve CAR T cell therapy in liquid and solid tumors.
Written by Gwen Cusing
Read more about this publication:
IL-9 as a naturally orthogonal cytokine with optimal JAK/STAT signaling for engineered T cell therapy. Jiang et al., Immunity (2026).
References:
[1] McDermott, Rn, Bsn, Ocn, Bmtcn, K. & Spendley, Agnp, Msn, Aocn, L. Anti-CD19 CAR T-Cell Therapy for Adult Patients With Refractory Large B-Cell Lymphoma. J. Adv. Pract. Oncol. 10, (2019).
[2] Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer J. Clin. 74, 229–263 (2024).
[3] Du, B. et al. CAR-T therapy in solid tumors. Cancer Cell 43, 665–679 (2025).
[4] Zhang, L. et al. Tumor-Infiltrating Lymphocytes Genetically Engineered with an Inducible Gene Encoding Interleukin-12 for the Immunotherapy of Metastatic Melanoma. Clin. Cancer Res. 21, 2278–2288 (2015).

Leave a comment