The success of nucleoside-modified mRNA vaccines has redefined modern vaccine platform design. These vaccines deliver antigen-encoding mRNA packaged inside lipid nanoparticles (LNPs), generating potent neutralizing antibodies and durable memory B cell responses. One defining feature of mRNA vaccination is its ability to drive strong germinal center (GC) reactions – the specialized structures in lymph nodes where high-affinity antibody-secreting cells are refined and long-lived memory B cells are born [1].
Yet despite their clinical impact, a basic mechanistic question has lingered: which part of the LNP-encased mRNA vaccine is responsible for programming these powerful GC responses?
LNPs are known to have intrinsic adjuvant activity. In contrast, nucleoside-modified mRNA has been described as immunologically “quiet,” carefully engineered to dampen excessive innate sensing. If that were entirely true, then the LNP should be doing most of the immunological heavy lifting.
In this study, Castaño et al. challenge that assumption and reveal a more nuanced picture: mRNA vaccines function as a coordinated two-component system in which both the mRNA and the LNP deliver distinct, complementary immune instructions [2].
The story begins with interferon.
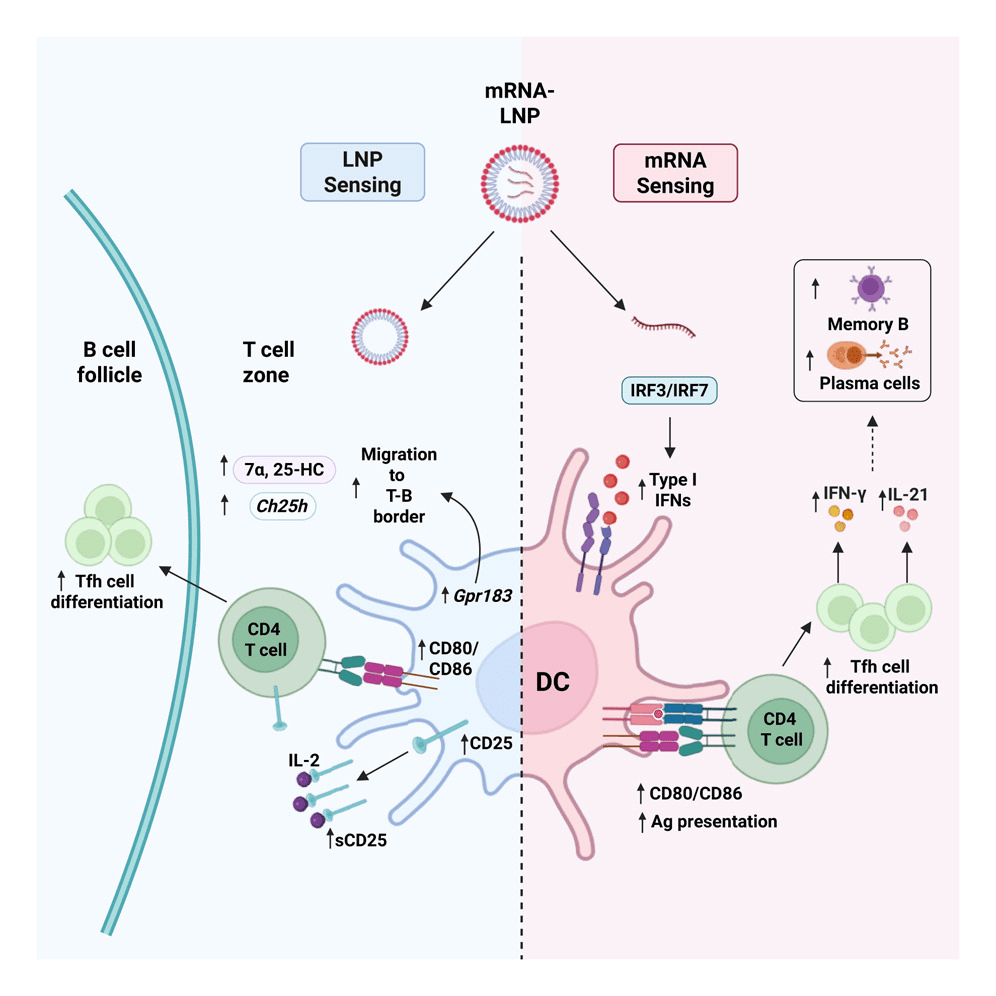
The authors show that mRNA-LNP vaccination induces measurable type I interferon (IFN-α/β) production in draining lymph nodes. When interferon signaling is blocked, particularly in dendritic cells (DCs), T follicular helper (Tfh) cell differentiation and GC B cell responses are significantly reduced. Because Tfh cells provide the essential help that drives germinal center selection and affinity maturation, this loss of interferon signaling ultimately limits the generation of high-affinity B cells. Importantly, this interferon dependence is much stronger when nucleoside-modified mRNA is present compared to protein delivered with empty LNPs.
This finding reframes the immunogenicity of modified mRNA. Rather than being fully silent, it provides a controlled interferon signal that enhances DC maturation and supports Tfh priming.
But interferon does more than increase the amount of cells present- it shapes quality. Tfh cells generated in the presence of modified mRNA produce more IFN-γ and IL-21, cytokines associated with antiviral helper function and effective B cell support. As a result, mRNA-containing vaccines generate stronger memory B cell responses and improve neutralization titers.
So, if modified mRNA supplies an increased interferon response, how is the LNP contributing to these GC responses?
Using single-cell transcriptional profiling of antigen-presenting DCs, the authors show that the dominant shift in DC gene expression is driven by the presence of LNPs. LNP-containing vaccines induce a broad activation and migration program in conventional type 2 DCs, which is the subset specialized for T follicular helper cell priming.
Among the LNP-induced pathways, two stand out as particularly relevant for Tfh differentiation: the IL-2 receptor α chain (CD25) and the chemotactic receptor Ebi2.
First, LNP exposure increases expression of CD25 on DCs. Because IL-2 signaling can suppress early Tfh commitment, CD25 expression allows DCs to act as an IL-2 sink, effectively lowering IL-2 availability in the local microenvironment. When CD25 is deleted specifically in DCs, Tfh and GC responses are diminished following mRNA vaccination.
Second, LNP administration promotes the production of the oxysterol ligand 7α,25-hydroxycholesterol by upregulating enzymes such as Ch25h. This ligand engages the receptor Ebi2, guiding immune cells toward the interface of the T cell zone and B cell follicle in the lymph node, a critical anatomical site for Tfh commitment. Disrupting this axis reduces Tfh responses and demonstrates that spatial positioning is not incidental but mechanistically important.
Taken together, these findings provide a mechanistic framework for understanding mRNA vaccine potency. The nucleoside-modified mRNA contributes a controlled type I interferon signal that enhances dendritic cell maturation and shapes Tfh functional quality, while the LNP component programs dendritic cells to regulate IL-2 availability and guide positioning within the lymph node. The potency of mRNA vaccines therefore emerges from the coordination of these distinct signals rather than from a single dominant adjuvant effect.
By decoupling these contributions, Castaño et al. provide a clearer framework for thinking about vaccine design. Tuning the interferon axis can influence helper quality, while adjusting nanoparticle composition may modulate dendritic cell activation and spatial organization. Understanding this division of labor offers a blueprint for tuning future vaccine platforms to optimize durability and immune response.
Written by Mandy Buck
Read more about this publication:
Distinct components of mRNA vaccines cooperate to instruct efficient germinal center responses. Castaño et al., Cell (2026).
References:
[1] Turner, Jackson S et al. “SARS-CoV-2 mRNA vaccines induce persistent human germinal centre responses.” Nature vol. 596,7870 (2021): 109-113. doi:10.1038/s41586-021-03738-2
[2] Castaño, Diana et al. “Distinct components of mRNA vaccines cooperate to instruct efficient germinal center responses.” Cell vol. 188,26 (2025): 7461-7480.e23. doi:10.1016/j.cell.2025.11.023

Leave a comment