Blood vessels seem remarkably simple; they are just tubes that carry blood throughout our body. The innermost layer of these vessels, however, is composed of a single, flat layer of specialized endothelial cells called the endothelium. These endothelial cells are incredibly dynamic as they have the capacity to “activate,” growing into new, small blood vessels. Most of the time, they are quiescent, an “inactive” state that allows endothelial cells to simply exist.
Endothelial cell activation and the subsequent blood vessel growth are referred to as angiogenesis. Signals that encourage angiogenesis can include oxygen deprivation in diseases like stroke, where blockages in blood vessels prevent blood flow to surrounding tissues. Endothelial cells can sense these low oxygen levels and activate genes that promote blood vessel growth. These new blood vessels can form new blood flow routes that bypass obstructions in older vessels. Andrade and colleagues study how endothelial cells control their quiescent, or dormant, state. A better understanding of this process could lead to new therapies for cardiovascular diseases.
YOU ARE WHAT YOU EAT
In its simplest form, metabolism is a set of chemical reactions that allows the body to change the food we eat into energy. Beyond energy, the breakdown of food can also create other small molecules (called metabolites) that can influence genes, proteins, and other reactions inside our bodies. One such metabolite is 2-hydroxyglutarate (2HG).
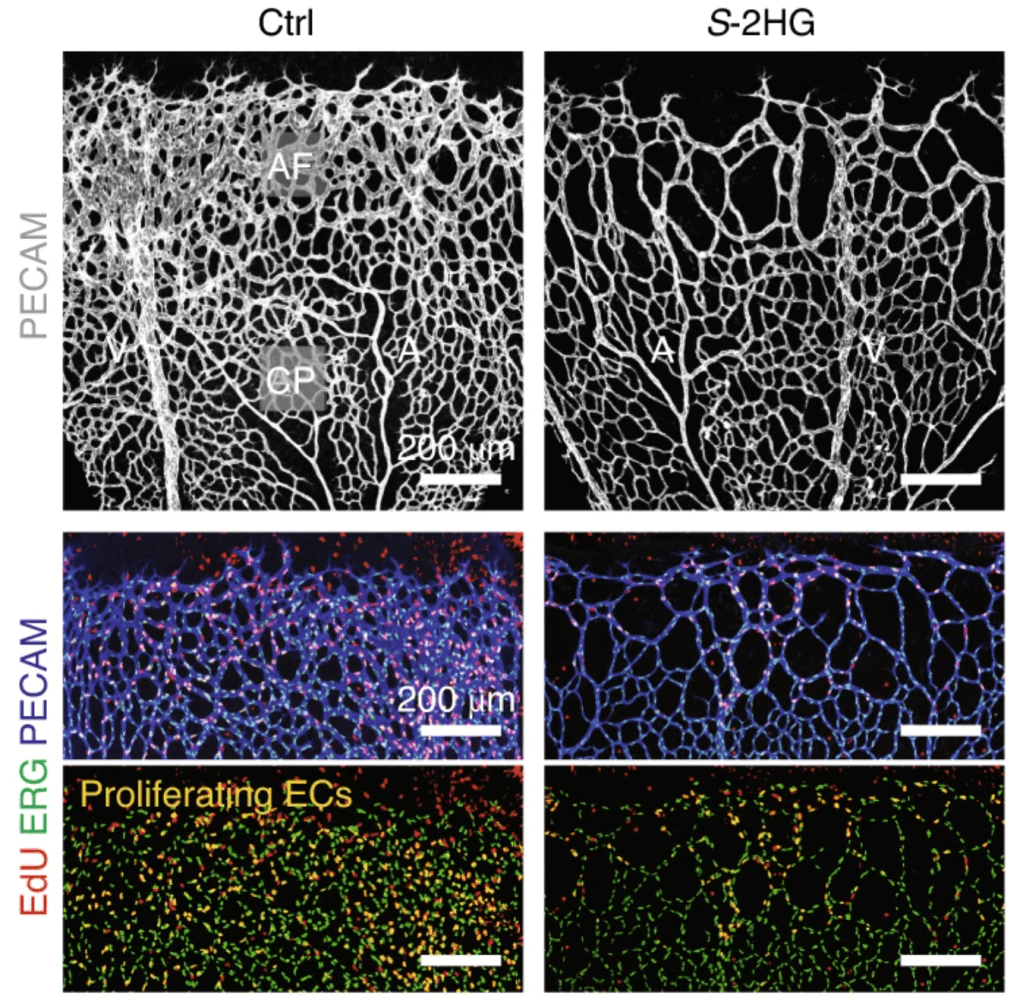
FOXO1 is well-established regulator of endothelial cell quiescence. In cells that have this gene perpetually turned on, Andrade et al. discovered that 2HG levels were increased [1]. When one form of this metabolite, S-2HG, was isolated and injected into developing mice eyes, there was clear impairment in the blood vessel network of the retina. It was clear that S-2HG was playing a role in controlling endothelial cell quiescence, and if levels of this metabolite are too high, it can prevent angiogenesis.
TOO MUCH OF A GOOD THING IS A BAD THING
Under normal conditions 2-oxoglutarate (2OG) is turned into succinyl-CoA, a reaction mediated by 2OG dehydrogenase (OGDH). But under specific conditions, 2OG can spontaneously turn into 2HG. By deleting OGDH in cells, very little 2OG was being converted into their normal product, and they could coax more production of 2HG instead. This created a scenario very similar to the “always on” FOXO1 cells, allowing them to connect how FOXO1 was specifically increasing the level of specific metabolites.
However, researchers failed to see any changes in the genes related to OGDH in the activated FOXO1 cells. If that were the case, could these two pathways be connected? Instead, they saw an increases in another family of metabolites: branched-chain amino acids (BCAAs). Interestingly, this family is known to specifically inhibit, or turn off, OGDH activity. It was clear that FOXO1 wasn’t directly influencing OGDH activity but rather producing small molecules that would mediate this activation and deactivation instead.
Together, these findings reveal a surprising link between metabolism and blood vessel growth. Rather than directly controlling enzymes involved in metabolism, FOXO1 controls small metabolites that indirectly shift how cells process nutrients. By increasing BCAAs, FOXO1 reduces OGDH activity, which allows more 2HG to accumulate. Higher levels of this metabolite keep endothelial cells in their quiescent state, preventing the formation of new blood vessels. Understanding this metabolic “brake” on angiogenesis could help scientists develop new therapies for diseases such as stroke and other cardiovascular diseases, where encouraging or limiting blood vessel growth may be beneficial.
Written by Caitlyn Dang
Read more about this publication:
Control of endothelial quiescence by FOXO1-regulated metabolites. Andrade et al., Nature Cell Biology (2021)
References
[1] DOI: 10.1038/s41556-021-00637-6

Leave a comment